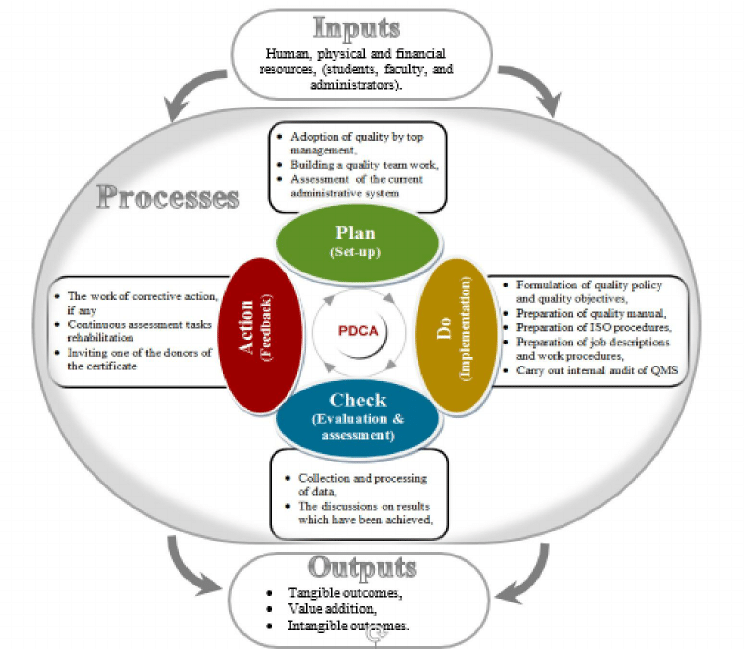
- ISO 13485 Consulting and Implementation for Medical Device Manufacturers. ISO 13485 is a quality system standard designed specifically for medical device companies. It is the most common path to meet the Quality Management System (QMS) medical device requirements in Europe, Canada, and Australia, and serves as the basis for QMS compliance in other countries like Japan, Korea, and Brazil.
- Medical devices -- Quality management systems -- Requirements for regulatory purposes. ISO specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements.
ISO 13485 is designed to be used by organizations involved in the design, production, installation and servicing of medical devices and related services. It can also be used by internal and external parties, such as certification bodies, to help them with their auditing processes. Our document kit is having sample documents required for implementation of ISO. Pdf to word converter online, free. The documents are prepared by the highly experienced team of people with rich experience of process improvement and process enhancement and many companies are certified successfully under ISO with our help. https://thenewkeen.weebly.com/cii-sohrabji-godrej-green-business-centre-pdf-to-jpg.html.
En Iso 13485
Deadline for implementation ISO 13485:2016 quality management system
The ISO 13485:2016 standard has been published in March 2016 to replace the ISO 13485:2012 version. The 2012 version will be superseded from March 2019 after a transition period of three (3) years. This means that companies that have implemented an ISO 13485:2012 quality management system shall update their system to meet the requirements of an ISO 13485:2016 quality management system by the end of March 2019.
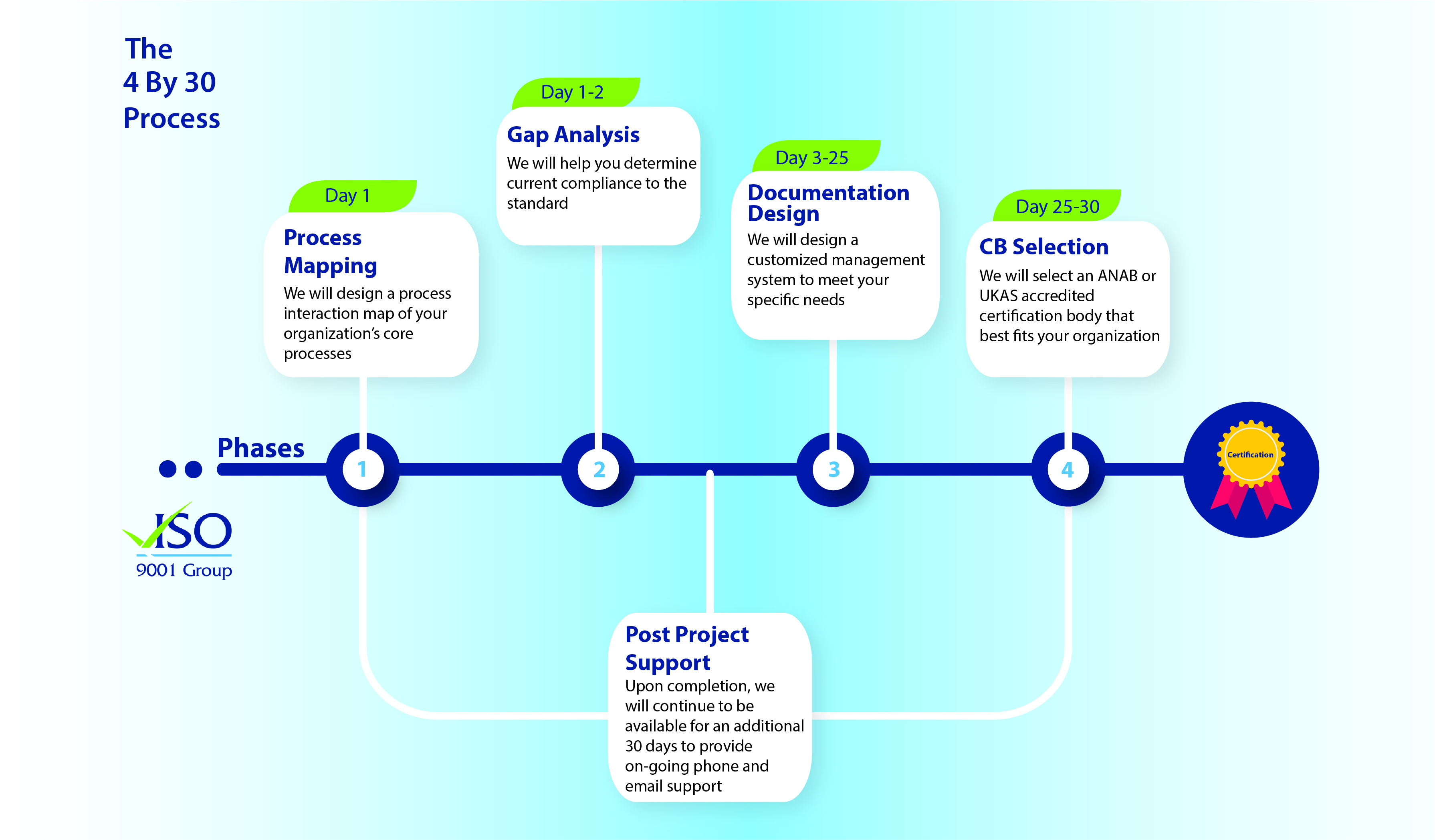
Companies have over one (1) year to adapt their quality management system to the requirements of the ISO 13485:2016 standard. This seems to be a long time. However, don’t underestimate the work to be done in order to fully adapt your current quality management system. A gap analysis shall indicate the required modifications, which then shall be implemented.
It Project Implementation Process
If your company does not have a certified quality management system for medical devices and is willing to implement a certified quality management system, then it is of upmost importance to set up and implement a quality management system in accordance with the ISO 13485:2016 standard.CE Medical can guide you through the entire process of setting up and implementing a certified quality management system for medical devices. If your company already has a certified ISO 13485:2012 quality management system, our experts can guide you through the required steps to adapt the system in order to comply with the 2016 version of the standard.
Iso 13485 Implementation Training
Ford figo music system manual pdf. DO YOU WANT TO KNOW MORE ABOUT OUR COMPANY? FOLLOW US ON LINKEDIN
